Discovery Site for Startups (5–7 pages)
Custom design, secure Next.js build, responsive layouts, contact/lead forms, and basic technical SEO. A fast, low-friction option for early-stage biotech and pharma ventures.

Specialist biotech website design and development for clinical, pharmaceutical, medical devices, and research organisations. We combine Next.js/React, Headless CMS, WordPress/Drupal options, and secure AWS hosting to create compliant, high-converting sites that showcase scientific services and generate investor leads online.
Trusted by 100+ Mumbai brands




Specialist biotech website design and development for clinical, pharmaceutical, medical devices, and research organisations. We combine Next.js/React, Headless CMS, WordPress/Drupal options, and secure AWS hosting to create compliant, high-converting sites that showcase scientific services and generate investor leads online.
Trusted by 100+ Mumbai brands





Our Approach
We design and build industry-focused websites for biotech, pharma, medical device, and laboratory teams. From UX strategy and visual systems to secure biotech website development—every project prioritises data privacy, regulatory compliance, and investor conversion.
Work with a team that understands clinical workflows, CRO lead generation, and how to showcase scientific services. We deliver responsive life sciences website design that supports clinical signups, partner outreach, and academic research communications.
Our builds are modular, documented, and production-ready: secure portals for labs, compliance-first patterns, and a content model that scales with your research and marketing needs.
How We Deliver High-Performing Biotech Websites
Discovery & Strategy
We map stakeholders—investors, clinicians, partners—and define pipelines that generate investor leads online, streamline clinical trial signups, and improve CRO lead generation.
UX/UI & Design System
Clinical-friendly UI, accessible patterns, and component libraries that present complex science clearly. Design systems make it easy to showcase scientific services and case studies.
Modern Tech Stack
Custom web development with Next.js/React, WordPress or Drupal options, and Headless CMS architectures. Secure AWS hosting and scalable APIs for lab portals and integrations.
Performance, Privacy & Compliance
Optimised performance plus data privacy, secure hosting, and regulatory compliance—so sites meet clinical and pharmaceutical standards while ranking well in search.
Content & CMS
Content models built for academic research, publications, and client testimonials. Editors can publish case studies, stock photos, and technical pages without breaking layouts.
Launch & Support
Staging, validation, analytics, and ongoing support. Maintenance plans include security patches, backups, and monitored hosting for data-sensitive projects.
Services
We offer end-to-end biotech web design: custom web development, biotech website packages, website redesign service, Headless CMS, WordPress and Drupal builds, secure AWS hosting, and CRO-focused UX to help you generate investor leads online and streamline clinical trial signups.
Choose a package built for life sciences teams. Every plan includes responsive design, secure hosting options, and analytics configured for clinical and investor funnels.
Add-ons cover SEO for academic research, CRO lead generation, gated assets, and integrations to CRM, EMR, or lab systems.
Custom design, secure Next.js build, responsive layouts, contact/lead forms, and basic technical SEO. A fast, low-friction option for early-stage biotech and pharma ventures.
IA/UX workshop, component library, gated assets, blog/CMS, and performance/SEO setup. Built to improve CRO lead generation and attract clinical partners.
Device catalogs, PDP/PLP templates, search/filters, secure payments, and analytics. Optimised for regulatory clarity and conversion for medical device suppliers.
UX audit, redesign, content migration, redirects, and Core Web Vitals uplift. Protect rankings and data privacy while you modernise.
Vitals tuning, schema for research, IA cleanup, and security hardening. Quick technical wins that compound organic growth and trust.
Monthly updates, monitoring, security patches, and secure AWS hosting. Keep your clinical or laboratory site compliant and reliable.
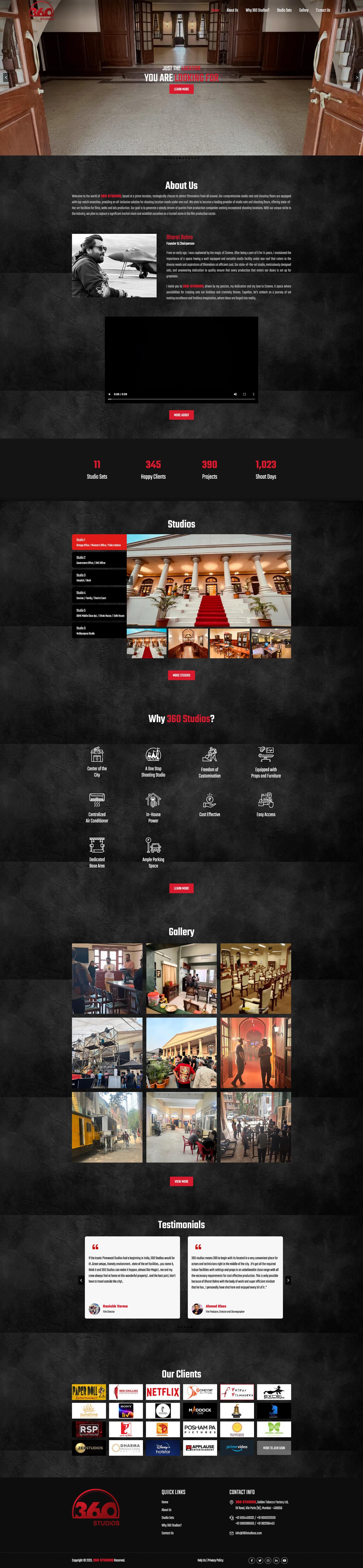
Creative studio website with bold visuals and portfolio showcase.
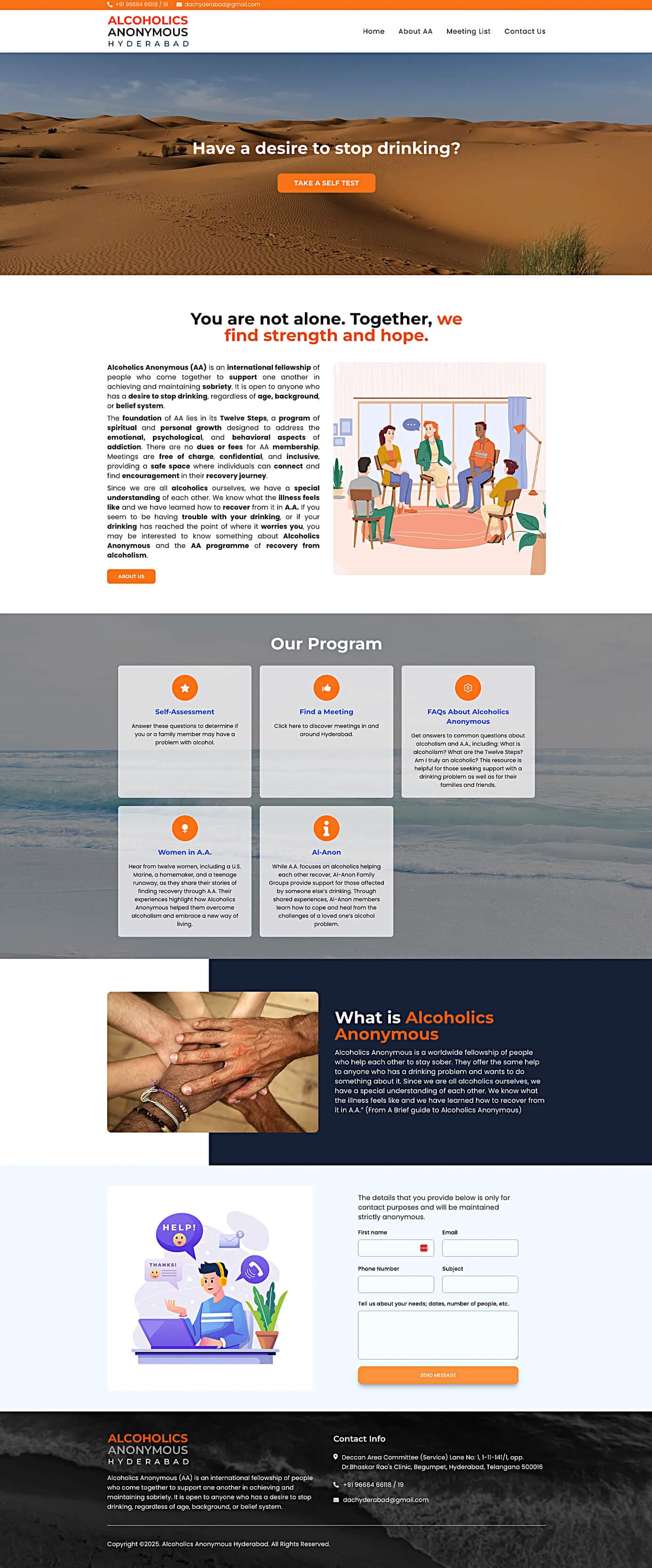
Corporate website for AA Hyderabad featuring meetings and event sections.

Professional website for Alturity Solutions technology services.

Industrial website with product catalogue, certifications, and contact forms.

Clean and modern architecture portfolio website.

Architecture firm website showcasing projects, careers, and office design.

360 Studios
Creative studio website with bold visuals and portfolio showcase.

AA Hyderabad
Corporate website for AA Hyderabad featuring meetings and event sections.

Alturity Solutions
Professional website for Alturity Solutions technology services.

Aniket Enterprises
Industrial website with product catalogue, certifications, and contact forms.

APARC
Clean and modern architecture portfolio website.

Archinova Inc.
Architecture firm website showcasing projects, careers, and office design.
READY WHEN YOU ARE
We provide a clear proposal, timeline, and a plan to generate investor leads online or streamline clinical trial signups. From UX to launch we prioritise privacy, performance, and compliance.
Expect fast responses, milestone check-ins, and production-ready code with integrations to CRMs and lab systems. Serving life sciences teams locally and globally.











FAQs
Direct answers to help you plan budgets, timelines, and compliance.
Need industry-specific examples? Tell us your focus—clinical, pharmaceutical, medical devices, or laboratory—and we’ll share relevant case studies and client testimonials.
Typical biotech and life sciences builds take 4–10 weeks depending on complexity, integrations, and regulatory reviews. A simple brochure site is faster; portals and CRM integrations require more time for security and testing.
Costs depend on scope: pages, custom web development, integrations, and compliance needs. We offer biotech website packages and will provide a transparent, fixed proposal after discovery.
Yes—we design with data privacy and regulatory compliance in mind. We can implement access controls, secure hosting on AWS, audit logs, and documentation to support clinical and pharmaceutical standards.
Absolutely. We connect to CRMs, marketing automation, and lab systems to streamline clinical trial signups, nurture investor leads online, and align data flows with privacy rules.
Yes. All projects include responsive design and testing across devices. Mobile-first patterns are especially important for field teams, conference attendees, and clinician recruitment.
Discovery, UX/UI, secure Next.js/React or WordPress/Drupal development, Headless CMS options, analytics, performance tuning, and hosting. We also provide guidance on content like academic research and lab case studies.
Yes—our website redesign service includes UX audits, content migrations, redirects, and a focus on maintaining search visibility while updating design and compliance features.
We design conversion funnels, gated investor decks, and landing pages to attract and qualify investor interest. Analytics and CRO experiments are included to improve lead quality.
We offer a mix of bespoke builds and starter templates. For teams that prefer DIY, we can supply templates and tutorial resources, but most biotech organisations opt for custom development to meet compliance needs.
You own the code and content after final delivery. We provide documentation, access credentials, and handover materials so your team can manage the site or continue with our maintenance plans.
Locations
We proudly serve clients in major cities worldwide, delivering exceptional web design, development, and digital marketing services.